Lithium
What is lithium?
 Lithium (chemical symbol: Li) is the lightest of all metals. It does not occur as a pure element in nature but is contained within mineral deposits or salts, including brine lakes and seawater. The contained concentration of lithium is generally low and there are only a limited number of resources where lithium can be economically extracted.
Lithium (chemical symbol: Li) is the lightest of all metals. It does not occur as a pure element in nature but is contained within mineral deposits or salts, including brine lakes and seawater. The contained concentration of lithium is generally low and there are only a limited number of resources where lithium can be economically extracted.
Lithium and its chemical compounds exhibit a broad range of beneficial properties including:
- The highest electrochemical potential amongst all metals.
- An extremely high co-efficient of thermal expansion.
- Fluxing and catalytic characteristics.
- Acts as a viscosity modifier in glass applications.
Chemical Applications:
Lithium can be processed to form a variety of chemicals including lithium carbonate, lithium bromide, lithium chloride, butyl lithium and lithium hydroxide. The fastest growing and second largest market for lithium globally is for use in batteries.
Other Applications:
- Rechargeable batteries for a variety of devices like mobile phones, laptops, digital camera and more utilize lithium.
- Certain non-rechargeable batteries for items like clocks and pacemakers also make use of the metal.
- Lithium metal can also to form alloys with aluminum or magnesium, which is used for armor plating and in aircraft, bicycles, and trains.
- Lithium carbonate is used in the field of making glass and ceramics. It is also involved in producing aluminum.
- Lithium stearate is used in cosmetics, plastics, and its grease is used in aircraft and marine applications as well as other areas.
Lithium use in Batteries:
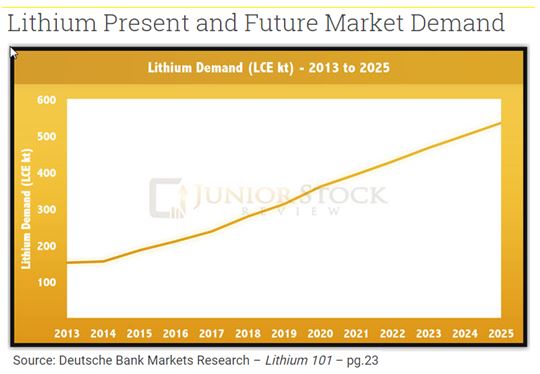
Lithium has a number of uses but one of the most valuable is as a component of high energy-density rechargeable lithium-ion batteries. Because of concerns over carbon dioxide footprint and increasing hydrocarbon fuel cost (reduced supply), lithium may become even more important in large batteries for powering all-electric and hybrid vehicles. It would take 1.4 to 3.0 kilograms of lithium equivalent (7.5 to 16.0 kilograms of lithium carbonate) to support a 40-mile trip in an electric vehicle before requiring recharge. This could create a large demand for lithium.
Batteries:
There are two types of lithium batteries,
Primary (non-Rechargeable): Including coin or cylindrical batteries used in calculators and digital cameras. Lithium batteries have higher energy density compared to alkaline batteries, as well as lower weight and long shelf and operating life.

Secondary (rechargeable): Key current applications for lithium batteries are in powering cell phones, laptops and other hand held electronic devices, power tools and large format batteries for electricity grid stabilisation. The advantage of the lithium secondary battery are its higher energy density and lighter weight, compared to nickel cadmium and nickel metal hydride batteries.
A growing application for lithium batteries is a power source for a wide range of electric vehicles, including electric bikes, scooters, buses, taxies, and passenger electric vehicles. There are three main categories of electric passenger vehicles; Hybrid Electric Vehicles, Plug-In Hybrid Vehicles and Electric Vehicles.


The electrification of vehicles is strongly supported by governments around the world due to increasing political and consumer focus on climate change and energy security. The introduction of mass produced passenger electric vehicles has the potential to significantly increase the future consumption of Lithium.
Other Chemical Applications:
Lithium chemicals are also used in a variety of other applications including:
- Lubricants: Lithium is used as a thickener in grease, ensuring lubrication properties are maintained over a broad range of temperature.
- Aluminum Smelting: the addition of lithium during aluminum smelting reduces power consumption, increases both electrical conductivity and reduces fluorine emission. The process increases strength and lightens the end product. These new aluminium/ lithium alloys are now being used in the auto and airline industry for fossil fuel savings.
- Air Treatment: Lithium may be used as an absorption medium for industrial refrigeration systems and for humidity control and drying systems.
- Pharmaceuticals: Lithium is used in the treatment for bi-polar disorder as well as in other pharmaceutical products.
Glass and Ceramics:
Glass: including container glass, flat glass, pharmaceutical glass, specialty glass and fiberglass. These glass products may be designed for durability or corrosion resistance or for the use at high temperatures where thermal shock resistance is important. The addition of lithium increases the glass melt rate, lowers viscosity and the melt temperature providing higher output energy savings and moulding benefits.
Ceramics: including ceramic bodies, frits glazes and heatproof ceramic cookware. Lithium lowers firing temperatures and thermal expansion and increases the strength of ceramic bodies. The addition of lithium to glazes improves viscosity for coating, as well as improving the glazes colour, strength and lustre.
Specialty Applications: including induction cook tops and cookware. Lithium’s extremely-efficient high of termalco expansion makes these products resistant to thermal shock and imparts mechanical strength.
Other Technical Applications:
Lithium is also used in a variety of metallurgical applications, including:
Steel Castings: the addition of lithium to continuous castings moulds fluxes assists in producing thermal insulation and lubricates the surface of the steel in the continuous casting process.
Iron Castings: in the production of iron castings, such as engine blocks, lithium reduces the effect of veining, thereby reducing the number of defective casts
Global Lithium Reserves
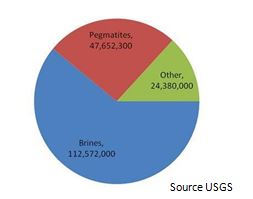
Global estimates suggest there are more than 30 million lithium resources, however, it is important to know that most deposits are not economically viable. For instance, some of the deposits, both brines and hard rock, may have high levels of impurities that make processing very costly, while others are in isolated parts of the world and would require high infrastructure expenditures, deeming them uneconomical. In the case of brines, the weather in some regions is not appropriate for the solar evaporation process. There are also many other factors, thus it is necessary to spend a significant amount of time and resources to determine the feasibility of these projects before considering them as viable resources (Euro Pacific-Canada, August 2013)
According to the United States Geological Survey (USGS) it is estimated that the global lithium reserves are in the 16 million tons. These estimates exclude lithium occurrences and resources that have not proven economic.
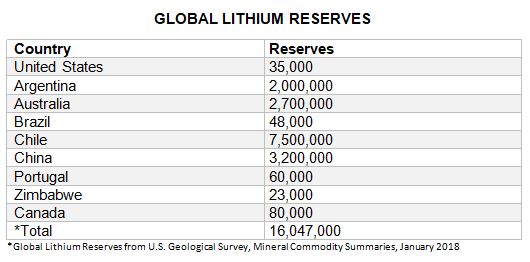
World Resources:
Owing to continuing exploration, lithium resources have increased substantially worldwide and total more than 53 million tons. Identified lithium resources in the United States, from continental brines, geothermal brines, hectorite, oilfield brines, and pegmatites, have been revised to 6.8 million tons. Identified lithium resources in other countries have been revised to approximately 47 million tons. Identified lithium resources in Argentina are 9.8 million tons; Bolivia, 9 million tons; Chile, 8.4 million tons; China, 7 million tons; Australia, 5 million tons; Canada, 1.9 million tons; Congo (Kinshasa), Russia, and Serbia, 1 million tons each; Czechia, 840,000 tons; Zimbabwe, 500,000 tons; Spain, 400,000 tons; Mali, 200,000 tons; Brazil and Mexico, 180,000 tons each; Portugal, 100,000 tons; and Austria, 50,000 tons.
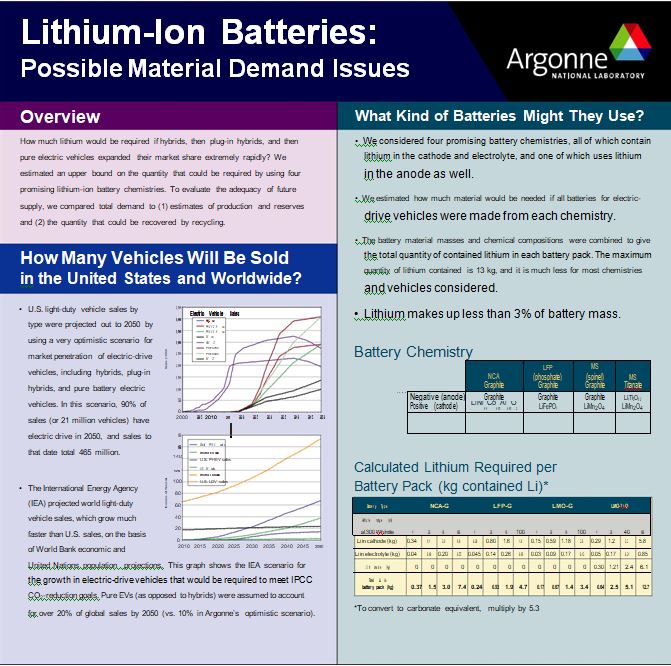
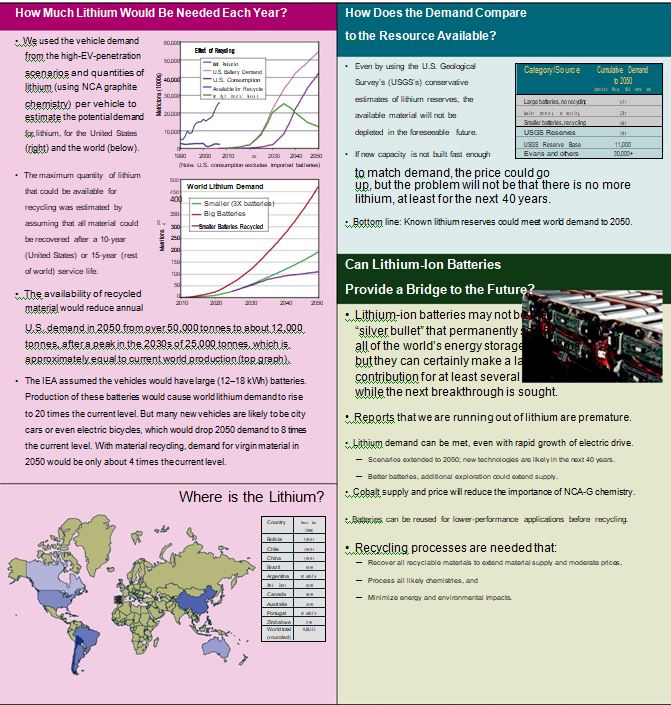
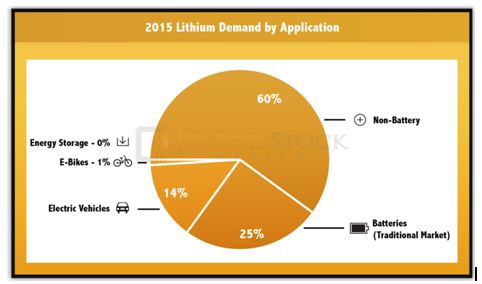
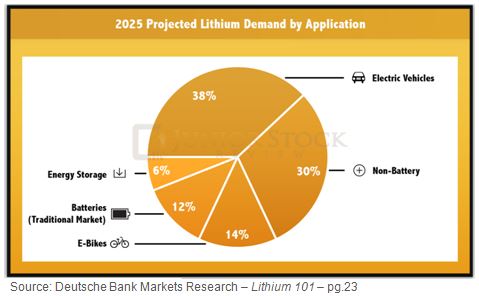
Lithium Demand by Application:
In this study below, we review the relevant geological features of the best characterized pegmatite
Pegmatites are commonly found throughout the world:
- These are course grained igneous rocks formed by the crystallization of post magmatic fluids.
- They occur in close proximity to large magmatic intrusions
- Lithium containing pegmatites are relatively rare and are most frequently associated with tin and tantalite.
- Lithium-rich granite pegmatites are much less common making up less than 1%
- Granite pegmatite-ore bodies are the hard-rock source of lithium
- The Lithium minerals that occur in granite pegmatites are
a) spodumene, (b) apatite, (c) lepidolite, (d) tourmaline and (e) amblygonite.
Lithium hard-rock recovery can be broken down into a few key steps:
1. crushing of the ore, then
2. concentration by froth floatation, followed by
3. hydrometallurgy and
4. precipitation from an aqueous solution. From here, depending on the application the manufacturer will create either
• Lithium hydroxide
• Lithium carbonate
which can be sent to factories to be manufactured into its final form.
When evaluating a hard-rock lithium deposit, the few key things to look for:
- Lithium Grade – The most important figure in any type of deposit. The higher the grade of lithium, the more economic the deposit.
- By-Products – can help reduce the cost per ton because they have value. Not to be confused with ‘harmful’ impurities,
- For lithium hard-rock deposits, tantalum, beryllium and caesium are examples of profitable by-products of the refinement process.
- Impurity Levels – Lower impurity levels will lead to increase their use in end use applications, such as glass and ceramics.
- Location - Access to good infrastructure is key to a profitable mining project
Images of Lithium after different processes:

For the sake of this narrative we are excluding the Brine based deposits as they are located all over the world but only a small number of them are considered to be economical for the recovery of lithium, potash, boron and potassium.
The Great Transition, Part I: From Fossil Fuels to Renewable Energy:
http://www.earth-policy.org/plan_b_updates/2012/update107